In the dynamic landscape of pharmaceutical manufacturing, the role of pharma intermediates is paramount. These compounds, essential building blocks in the synthesis of active pharmaceutical ingredients (APIs), dictate the quality, efficacy, and safety of final drug products. The global pharmaceutical industry is experiencing unprecedented growth, driven by an aging population, increasing prevalence of chronic diseases, and continuous innovation in drug discovery. This escalating demand places immense pressure on the supply chain for high-quality chemical precursors, emphasizing the need for reliable, compliant, and technically advanced suppliers.
Key industry trends underscore this evolution. Firstly, there's a pronounced shift towards specialized and complex molecules, requiring bespoke synthesis routes and highly purified intermediates. Secondly, regulatory scrutiny, particularly from bodies like the FDA and EMA, is intensifying, demanding rigorous adherence to cGMP (current Good Manufacturing Practices) and comprehensive documentation throughout the supply chain. Thirdly, sustainability and green chemistry principles are gaining traction, pushing manufacturers to adopt environmentally friendly processes and materials. Compounds like N,N’-Dimethyl Urea, often utilized as a crucial reagent or intermediate, exemplify the precision required in this sector, serving as precursors for a range of pharmaceutical compounds and sometimes even as an intermediate in specialized polymer applications as additives for polymers. Our focus here is to provide B2B decision-makers and engineers with a comprehensive understanding of the value proposition offered by meticulously produced pharma intermediates.
The production of essential pharmaceutical building blocks is a complex, multi-stage process demanding absolute precision, stringent quality control, and robust adherence to pharmaceutical standards. Our manufacturing methodology ensures the highest purity and consistency, critical for downstream API synthesis.

Figure 1: Schematic representation of a typical manufacturing facility for pharmaceutical intermediates.
Our high-purity chemical intermediates serve a broad spectrum of industries beyond traditional pharmaceuticals, including specialized chemical synthesis and advanced materials.
Advantages in these scenarios include enhanced product performance, energy saving during client's downstream processing due to optimized intermediate properties, and superior corrosion resistance of the final products derived from our intermediates, leading to extended shelf-life and stability.
Refine N,N’-Dimethyl Urea (DMU) is a prime example of a versatile pharma intermediates, crucial in various organic synthesis routes. Its high purity and consistent quality make it indispensable for demanding pharmaceutical and specialty chemical applications.
| Parameter | Specification | Method |
|---|---|---|
| Chemical Name | N,N’-Dimethyl Urea | Internal Standard |
| CAS Number | 96-31-1 | |
| Molecular Formula | C3H8N2O | |
| Molecular Weight | 88.11 g/mol | |
| Appearance | White Crystalline Powder | Visual |
| Purity (Assay) | ≥ 99.5% (HPLC) | HPLC |
| Melting Point | 105-108 °C | USP <741> |
| Moisture Content | ≤ 0.5% | Karl Fischer |
| Residue on Ignition | ≤ 0.1% | USP <281> |
| Heavy Metals (Pb) | ≤ 10 ppm | ICP-MS |
| Solubility | Soluble in water, ethanol, chloroform |
These stringent specifications ensure that our Refine N,N’-Dimethyl Urea meets the exacting requirements for pharmaceutical synthesis, contributing to the development of safe and effective medications. The low impurity profile and high assay are critical for minimizing purification steps in API manufacturing and preventing potential side reactions.
The versatility of high-quality pharmaceutical building blocks like Refine N,N’-Dimethyl Urea extends across a wide array of pharmaceutical and industrial applications. Their reliable chemical properties and purity are indispensable for various synthesis pathways.
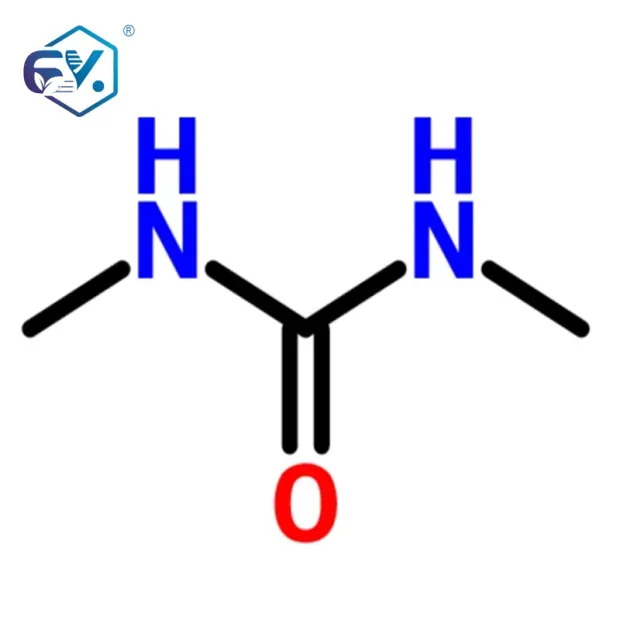
Figure 2: Diverse application landscape for high-quality chemical intermediates across pharmaceutical and industrial sectors.
The breadth of these applications highlights why a consistent supply of meticulously manufactured pharmaceutical precursors is not just beneficial, but absolutely vital for innovation and production across multiple high-tech industries.
Investing in high-quality pharma intermediates offers substantial technical and economic advantages for pharmaceutical manufacturers and specialty chemical producers. Our commitment to excellence translates into tangible benefits for our partners.
By choosing our premium pharmaceutical precursors, partners gain not just a chemical compound, but a comprehensive solution that reduces risks, optimizes processes, and ultimately contributes to the successful and timely delivery of critical drug products to market. The technical superiority of our products, such as Refine N,N’-Dimethyl Urea, ensures maximum value at every stage of the pharmaceutical value chain.
Selecting a supplier for critical pharmaceutical chemical building blocks is a strategic decision that impacts the entire drug development and manufacturing lifecycle. It goes beyond mere price points; it encompasses quality, reliability, and technical partnership.
| Feature/Parameter | Premium Supplier (e.g., HBGX Chemical) | Standard Supplier |
|---|---|---|
| Purity (N,N’-Dimethyl Urea) | ≥ 99.5% (HPLC, consistent batch-to-batch) | ≥ 98.0% (variable purity, higher impurities) |
| Impurity Profile | Extremely low and well-characterized; no problematic contaminants | Higher levels, less characterized; potential for unknown impurities |
| Regulatory Documentation | Comprehensive cGMP, ICH Q7, DMF support, full audit access | Basic COA, limited regulatory support |
| Supply Chain Security | Multiple qualified raw material sources, robust inventory management, global logistics | Single source dependence, limited inventory, local logistics only |
| Technical Support | Dedicated team of chemists, process optimization assistance, troubleshooting | Limited to sales support, no scientific guidance |
| Price Point (relative) | Higher initial cost, but lower total cost of ownership (TCO) due to reduced risks and processing. | Lower initial cost, but higher TCO due to quality issues, re-work, and regulatory hurdles. |
The choice of a premium supplier, despite a potentially higher upfront cost, ultimately leads to significant savings and reduced risks across the entire pharmaceutical value chain. Our commitment to excellence, coupled with robust quality systems and unparalleled technical support, positions us as the preferred partner for critical pharmaceutical manufacturing components.
Recognizing that every pharmaceutical project has unique requirements, we offer highly customized solutions for chemical intermediates. Our approach centers on deep collaboration to meet specific technical, regulatory, and logistical needs.
Our commitment to customized solutions solidifies our role as a strategic partner, not just a supplier. By addressing specific challenges and aligning with client objectives, we help accelerate drug development cycles and enhance overall product quality.
Our expertise in providing high-quality pharmaceutical precursors is demonstrated through successful collaborations with leading pharmaceutical companies worldwide. These case studies highlight our ability to deliver critical materials that accelerate drug development and ensure product integrity.
A global pharmaceutical company was developing a novel oncology API that required a highly specialized and enantiomerically pure intermediate. Their previous supplier struggled with batch-to-batch consistency and high levels of a critical impurity, leading to significant delays in clinical trials. We engaged with their R&D team, providing a high-purity chemical intermediate synthesized using an optimized, proprietary route. Our product consistently exceeded their purity requirements (≥ 99.8% enantiomeric excess and <0.05% critical impurity), demonstrating exceptional batch uniformity. This not only enabled the client to resume their clinical trials promptly but also reduced their purification steps for the API by 30%, resulting in substantial cost savings and an expedited path to regulatory submission. This successful partnership underscored our capability in complex synthesis and stringent quality control.
A major veterinary pharmaceutical manufacturer faced challenges with the stability and bioavailability of a broad-spectrum antibiotic. Investigation traced the issue back to a trace impurity in their dimethylurea supply, which was acting as a catalyst for degradation over time. Upon switching to our Refine N,N’-Dimethyl Urea, characterized by its exceptionally low impurity profile and controlled moisture content, the client observed a remarkable improvement. The degradation rate of their antibiotic formulation decreased by 50% over a 24-month stability study. This enhanced stability translated into an extended shelf-life for their final product, reduced recall risks, and ultimately, improved efficacy and trust among veterinary practitioners. This highlights how the superior quality of a fundamental intermediate like dimethylurea can have a profound impact on downstream product performance and market success.
At the core of our operations is a steadfast commitment to client trust, backed by transparent practices and comprehensive support services. We understand that reliability is paramount in the pharmaceutical industry.
Q: What quality certifications do you hold for your pharmaceutical intermediates?
A: Our manufacturing facilities are ISO 9001:2015 certified, and all relevant products are produced under cGMP guidelines. We also ensure compliance with specific pharmacopoeia standards (USP, EP, JP) and provide comprehensive documentation including Certificates of Analysis (CoA), MSDS, and support for Drug Master Files (DMFs).
Q: Can you accommodate custom synthesis requests for specific intermediates?
A: Yes, we specialize in custom synthesis and process development. Our R&D team works closely with clients from laboratory scale to commercial production, ensuring tailored solutions for unique molecular structures or specific purity profiles.
Q: What is your typical lead time for orders?
A: Lead times vary depending on product availability, quantity, and specific customization requirements. For standard products like Refine N,N’-Dimethyl Urea, we typically fulfill orders within 2-4 weeks. Custom synthesis projects will have a project-specific timeline communicated upfront.
Q: What kind of after-sales support do you provide?
A: We offer extensive after-sales support, including dedicated technical assistance from our team of chemists, regulatory guidance, and responsive customer service for any logistics or product-related inquiries. We are committed to resolving any issues swiftly and efficiently.
Our robust supply chain and optimized production planning enable efficient order fulfillment. Standard bulk orders for high-demand pharmaceutical intermediates are typically processed and ready for shipment within 14-28 calendar days. For urgent requirements or specialized products, expedited services can be arranged. We leverage global logistics networks to ensure timely and secure delivery to your facility, with full traceability from our plant to your dock.
We stand by the quality of our chemical building blocks. All products are warranted to meet or exceed the specifications provided in their respective Certificates of Analysis (CoA) and comply with agreed-upon pharmacopoeial standards at the time of shipment. In the rare event of a product not meeting these specifications, we are committed to prompt investigation, replacement, or credit, in accordance with our quality assurance protocols and mutual agreement. Our quality management system includes rigorous batch retention and testing to support any investigations.
Our dedicated customer support team and technical experts are available to assist you throughout your journey. From initial inquiry and technical consultation to order placement and post-delivery support, we ensure a seamless and responsive experience. Contact us via our website, email, or direct phone lines for expert assistance. We pride ourselves on building long-term, trust-based relationships with our clients, underpinned by reliable product quality and exceptional service.
The demand for high-purity pharma intermediates continues to grow, reflecting the increasing complexity and regulatory demands of the global pharmaceutical industry. As a dedicated provider, we are committed to delivering superior quality, technical excellence, and unwavering support to our partners. Our Refine N,N’-Dimethyl Urea exemplifies this commitment, offering unmatched purity and consistency vital for critical applications. By partnering with us, pharmaceutical manufacturers can ensure the integrity of their supply chain, accelerate drug development, and confidently bring safe and effective medicines to market.
Understanding Pharmaceutical Intermediates Meaning Simply.
If you are interested in our products, you can choose to leave your information here, and we will be in touch with you shortly.