The global market for advanced antimicrobial agents continues its robust expansion, driven by escalating healthcare-associated infections (HAIs), growing public health awareness, and stringent regulatory mandates. Among these critical compounds, Chlorhexidine Gluconate (CHG) stands out as a cornerstone antiseptic, vital across numerous sectors. Recent market analyses project a significant compound annual growth rate (CAGR) for the CHG market, reflecting its indispensable role in infection control strategies.
Key drivers include the increasing adoption of CHG-based products in surgical preparation, wound care, and oral hygiene, particularly in developing economies. Furthermore, the veterinary sector and general industrial disinfection applications are witnessing expanded usage due to CHG’s broad-spectrum efficacy against gram-positive and gram-negative bacteria, fungi, and certain viruses. The shift towards preventive healthcare and the demand for advanced antiseptic solutions in both clinical and community settings further solidify CHG's market position.
Regulatory bodies worldwide, such as the FDA and EMA, continually underscore the importance of proven antimicrobial agents, fostering a market environment where high-purity, well-characterized Chlorhexidine Gluconate is highly sought after. Innovations in formulation, including alcohol-free and slow-release options, are also contributing to market diversification and addressing specific application needs.
Understanding the precise technical specifications of Chlorhexidine Gluconate is paramount for its effective and safe application in pharmaceutical, medical, and personal care products. Our high-grade CHG solutions are meticulously manufactured to meet and often exceed international pharmacopeial standards such as USP, EP, and BP.
| Parameter | Specification | Test Method / Standard |
|---|---|---|
| Aparência | Clear, almost colorless or pale yellow liquid | Visual |
| Concentration (w/v) | 19.0% - 21.0% (as Chlorhexidine Gluconate) | Titration (USP/EP method) |
| Identification | Conforms to reference spectrum | HPLC, UV Spectroscopy |
| pH (1% solution) | 5.5 - 7.0 | pH Meter |
| Specific Gravity (20°C) | 1.06 - 1.07 | Pycnometer |
| Related Substances | Total impurities < 2.0% | HPLC |
| Chloraniline (p-Chloroaniline) | < 50 ppm | HPLC |
| Microbial Count (Total Aerobic) | < 100 CFU/ml | USP <61> |
| Endotoxins | < 0.5 EU/ml | LAL Test (USP <85>) |
These parameters ensure that our Chlorhexidine Gluconate products are of the highest quality, suitable for direct incorporation into regulated pharmaceutical and medical device formulations where purity, stability, and efficacy are non-negotiable.
The synthesis of Chlorhexidine Gluconate is a complex, multi-stage chemical process demanding stringent control over reaction parameters, material purity, and environmental conditions to yield a high-quality pharmaceutical-grade product. Our manufacturing process adheres strictly to Good Manufacturing Practices (GMP) and ISO 9001 quality management systems, ensuring exceptional batch consistency and purity.
High-purity p-chloroaniline, dicyandiamide, and gluconic acid are sourced from qualified suppliers. Each raw material undergoes rigorous analytical testing (e.g., FTIR, HPLC, titration) to confirm identity, purity, and absence of critical impurities, complying with relevant ANSI/ASTM standards.
Dicyandiamide reacts with hydrogen chloride to form an intermediate compound, biguanide. This reaction is carefully controlled for temperature and pH to optimize yield and prevent side reactions. In-process control checks ensure reaction completion.
The biguanide intermediate then undergoes condensation with p-chloroaniline, typically in a solvent, at elevated temperatures. This crucial step forms Chlorhexidine base. Reaction kinetics are monitored by HPLC to ensure high conversion and minimize impurity formation.
The crude Chlorhexidine base is subjected to multiple purification steps, including solvent extraction, recrystallization, and filtration. This removes unreacted raw materials, by-products, and other contaminants, enhancing the overall purity to meet pharmaceutical specifications.
The purified Chlorhexidine base is reacted with gluconic acid to form the more soluble Chlorhexidine Gluconate salt. This process requires precise stoichiometric control and pH adjustment to ensure complete conversion and optimal salt formation. The solution is then clarified.
The CHG solution undergoes sterile filtration to remove particulate matter and ensure microbial purity, meeting USP <797> or equivalent standards. It is then aseptically filled into designated container111s, sealed, and prepared for shipment. Each batch undergoes final quality control testing against pharmacopeial specifications.
Throughout this entire process, advanced process control systems (e.g., DCS, PLC-based) monitor critical parameters such as temperature, pressure, flow rates, and concentrations. This meticulous approach ensures not only product efficacy but also environmental safety and optimal resource utilization, contributing to an extended service life in downstream products due to exceptional stability and purity. Our CHG is designed for industries like pharmaceuticals, medical devices, and personal care, where corrosion resistance of handling equipment is a concern and the active ingredient's stability is paramount.
The versatility and potent antimicrobial properties of Chlorhexidine Gluconate make it indispensable across a diverse range of critical industries. Its applications span from human healthcare to veterinary medicine, ensuring high standards of hygiene and infection control.
The demonstrated efficacy in these varied fields underscores CHG’s critical role in public health and safety protocols globally. Its ability to provide persistent antimicrobial action on surfaces and skin is a significant advantage in reducing pathogen transmission.
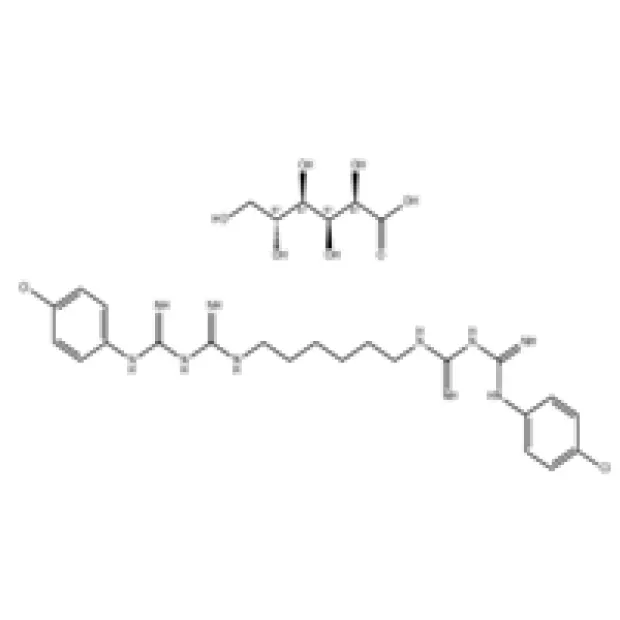
Figure 1: High-purity Chlorhexidine Gluconate solution ready for pharmaceutical applications.
Our Chlorhexidine Gluconate distinguishes itself through a suite of technical advantages that directly translate into superior performance and reliability for our B2B partners. These benefits are derived from our stringent manufacturing controls, advanced purification techniques, and commitment to quality.
These intrinsic properties, combined with our rigorous quality control, position our CHG as a premium choice for manufacturers seeking reliable, high-performance antimicrobial ingredients for their mission-critical products. The result is often an energy saving through optimized processes and reduced contamination risks, alongside a robust resistance to degradation even in challenging industrial environments.
Selecting the right supplier for Chlorhexidine Gluconate is a critical strategic decision that impacts product quality, regulatory compliance, and supply chain reliability. While numerous vendors exist, distinctions in manufacturing practices, quality assurance, and customer support are significant.
| Attribute | HBGX Chemical (Our Strengths) | Typical Competitor |
|---|---|---|
| Purity & Impurity Profile | >99.5% purity, p-chloroaniline < 50 ppm, low related substances. Fully compliant with USP/EP/BP. | Often <99% purity, p-chloroaniline > 100 ppm, variable related substances. May meet basic pharmacopeial standards but lack superior purity. |
| Manufacturing Standards | GMP-certified facilities, ISO 9001, rigorous IPCs, dedicated quality control labs with advanced analytical equipment. | May have basic certifications; inconsistent IPCs, often rely on external labs or less advanced in-house testing. |
| Technical Support & Expertise | Dedicated team of chemists and regulatory specialists. Comprehensive documentation, formulation guidance, and rapid issue resolution. Years of experience in B2B chemical supply. | Limited technical support, often basic COAs, minimal formulation advice, and longer response times. |
| Supply Chain & Reliability | Robust, resilient global supply chain with multiple sourcing options for raw materials. Excellent on-time delivery record. Transparent lead times. | Vulnerable supply chain, potential for delays, less transparent regarding material origins and logistics. |
| Customization Capabilities | Flexible manufacturing for tailored concentrations, packaging, and specific documentation needs. Collaborative R&D for novel applications. | Standard product offerings only, limited or no ability to customize batches. |
HBGX Chemical's commitment to superior product quality, transparent operations, and comprehensive customer support positions us as a trusted partner. Our proactive approach to regulatory compliance and deep technical expertise ensure that your products incorporating Chlorhexidine Gluconate will meet the highest industry benchmarks, fostering long-term success and market confidence.
Recognizing that diverse applications often require unique specifications, HBGX Chemical offers a robust suite of customized solutions for Chlorhexidine Gluconate. Our in-house R&D capabilities and flexible manufacturing infrastructure allow us to tailor our offerings precisely to our clients' distinct needs, ensuring optimal performance and seamless integration into their product lines.
Our collaborative approach ensures that you receive a CHG solution that is not merely a commodity, but a precisely engineered component designed to enhance the quality, efficacy, and market competitiveness of your final product. We partner with you to overcome formulation challenges and meet unique market demands.
Our Chlorhexidine Gluconate has been successfully integrated into a multitude of critical applications, demonstrating tangible benefits in real-world scenarios. These case studies highlight our product's performance and our commitment to client success.
A major hospital network integrated our 2% Chlorhexidine Gluconate solution with 70% isopropyl alcohol for pre-surgical skin preparation across all surgical departments. Over a 12-month period, detailed infection control data was collected and analyzed.
A leading dental care product manufacturer sought to reformulate their antiseptic mouthwash to improve efficacy and achieve a longer shelf life while complying with strict FDA oral care guidelines. They partnered with us for a custom-concentrated Chlorhexidine Gluconate solution.
A large veterinary hospital chain implemented a new disinfection protocol utilizing a 0.5% aqueous solution of our CHG for routine surface disinfection in examination rooms, surgical suites, and kennels.
A1: Our Chlorhexidine Gluconate solution should be stored in tightly closed container111s, protected from light, at room temperature (typically 15-30°C). Avoid freezing. Proper storage ensures its stability and prevents degradation over its warranted shelf life.
A2: When stored under recommended conditions, our standard 20% Chlorhexidine Gluconate solution has a typical shelf life of 36 months from the date of manufacture. Detailed expiration dates are provided on the Certificate of Analysis (CoA) for each batch.
A3: Yes, our CHG undergoes terminal sterile filtration processes, and we offer grades suitable for sterile applications. We provide relevant documentation including bioburden testing and endotoxin levels, ensuring compliance with pharmacopeial standards for sterile ingredients.
A4: We employ advanced purification techniques and strict control over reaction parameters during the synthesis process. High-performance liquid chromatography (HPLC) is used for in-process monitoring and final product release testing to ensure p-chloroaniline levels are consistently well below the regulatory limits (e.g., <50 ppm).
HBGX Chemical is committed to efficient and reliable order fulfillment for our Chlorhexidine Gluconate products. Our optimized logistics network and robust inventory management ensure timely delivery to meet your production schedules.
For large volume orders or recurring supply agreements, we encourage establishing a long-term supply plan to ensure continuous availability and optimized pricing.
At HBGX Chemical, we stand behind the quality and performance of our Chlorhexidine Gluconate with clear and comprehensive warranty commitments:
Our aim is to build long-term relationships based on trust and consistent product excellence. For detailed warranty statements, please refer to our standard sales agreements or contact our customer service team.
HBGX Chemical provides unparalleled customer support and after-sales service to ensure our partners derive maximum value from our Chlorhexidine Gluconate products. Our commitment extends beyond delivery, fostering a partnership approach to your success.
For immediate assistance, please visit our contact page at www.hbgxchemical.com/contact.html or reach out to your dedicated account manager. We are here to support your success every step of the way.
Buparvaquone-CAS 88426-33-9|Pharmaceutical Applications&Chemical Synthesis
Se você estiver interessado em nossos produtos, deixe suas informações aqui e entraremos em contato com você em breve.