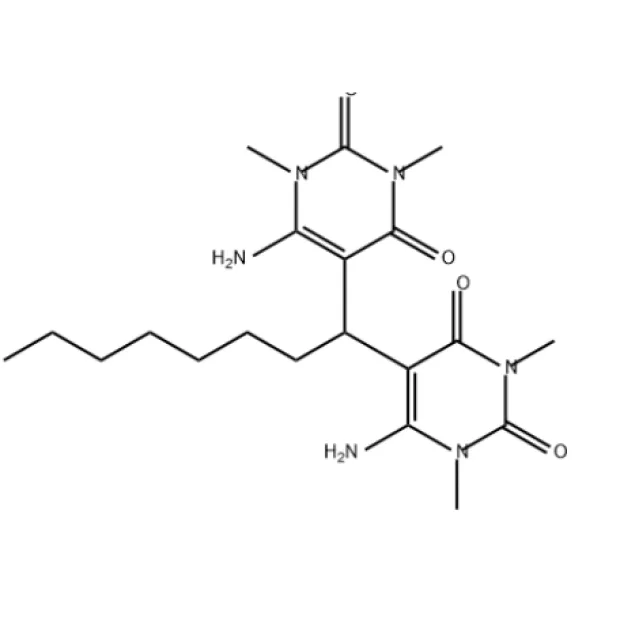
The US market is currently witnessing a massive shift toward domestic reshoring of pharmaceutical supply chains. There is an intensified demand for reliable uracil production to support oncology and antiviral research hubs across the East Coast and Midwest.
Simultaneously, the construction and automotive sectors in North America are upgrading their material standards. The transition toward non-toxic, high-efficiency pvc stabilizer systems is driven by strict EPA regulations and the push for sustainable building materials in urban developments.
Logistical challenges and the volatility of raw material costs have forced US manufacturers to seek partners who can optimize complex processes, such as the efficient uracil synthesis from urea, to reduce waste and lower the total cost of ownership.